Treatment and Distribution
FACTOID: Poor water sanitation and lack of safe drinking water kill more people each year than war, terrorism, and weapons of mass destruction combined.
Get a glass of water out of the nearest faucet. Now take a drink.
Is the water sparkling clear? Did it taste OK? Did it make you sick?
The answers to this should be yes, yes and no. And for most of us in the U.S. and Australia, which have some of the safest drinking water in the world, it is. But for 4 out of 10 people in the world, the answer is no, no and yes and there is a very good chance that the water that you drink, or bath in, or cook with, will make you sick.
FACTOID: The Center for Disease Control and the National Academy of Engineers have named water treatment as one of the most significant advances of the 20th century.
So, why is your water safe to drink? Because it's treated using chemicals and physics to make it safe. How it's treated and how much it's treated depends on its original source.
Water treatment isn't new, but how old is it? Well, the ancient Greeks invented more than the Olympics, democracy, and togas. They were among the first civilizations to move water from place to place using aquaducts (water distribution) and develop methods to improve the safety of drinking water (water treatment). Although they didn't know the causes of 'bad' water that made people ill, they did know that clear water was healthier to drink than cloudy water. Some of the methods they developed to make water clear (and thus safer to drink) were filtering water through charcoal, exposing it to sunlight, boiling it and staining it.
TOPIC OF DISCUSSION: Why do you think cloudy water could be hazardous to your health?

THE 1854 BROAD STREET PUMP CHOLERA EPIDEMIC
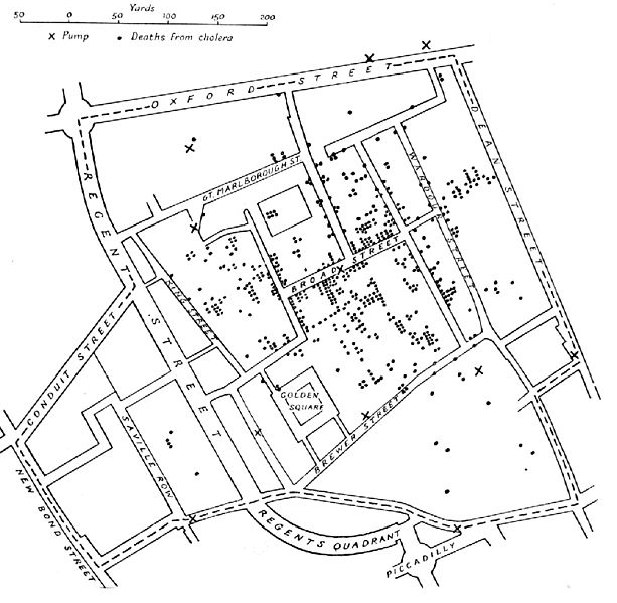 Original map by John Snow showing the clusters of cholera cases in the London epidemic of 1854.
Original map by John Snow showing the clusters of cholera cases in the London epidemic of 1854.Where does your drinking water originate?
Do you know? A reservoir? Groundwater? A river? Call your local water district or water treatment plant and ask them. Have their every been incidents of health concern in your local area with quality of water supply? If so, what caused these issues?
The source of water is important because water may be treated differently depending on where it comes from. That water coming out of your tap can originate from any one of many different water sources including groundwater, rivers, streams, reservoirs, rainfall and even the ocean. Groundwater tends to be cleaner than water from surface sources such as rivers and reservoirs which have been exposed to sediment and pollution, so it requires less treatment to be used as safe drinking water. Well water is groundwater, which is why it tends to have less treatment than public water sources. Does your school have a well? Do any of the students in your class have wells? Given the amount of water high schools use, the odds are likely that you get your high school water from a public water supply. If you use less water, such as at home, or are far away from public water sources such as on an island, then it's likely that you have a well.
FACTOID: Alum is also used in baking powders for cooking, as a flame retardant, and as an underarm deodorant.
So how is water treated to make it safe to drink? Water treatment is a broad term that describes the industrial scale processes of making water safe for a variety of uses, which includes drinking water, water for industry, medical and many other uses. The first step in treatment is coagulation. A chemical (usually Alum, also known as hydrated potassium aluminium sulfate, or more affectionately, KAl(SO4)2·12H2O is added to the water. Alum's negative charge attracts the positively charged dirt and bigger particles in the water and together they form larger particles called floc.
TOPIC OF DISCUSSION: What is the difference between "soft" and "hard" water?
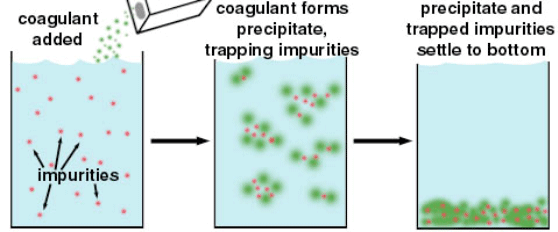 Process of adding coagulant to settle impurities to the bottom
Process of adding coagulant to settle impurities to the bottomTOPIC OF DISCUSSION: What causes distribution pipes to corrode?
During the next treatment stage, sedimentation, the heavier (than water) floc settles to the bottom and the clearer water is removed from the surface. This clearer water is then passed through a series of filters in a filtration step. Water filters take a variety of forms. They can be made of sand, gravel, charcoal or membranes. They can also have different pore sizes to remove different sized particles that were not removed by the coagulation step, such as dust, parasites, bacteria, viruses and chemicals. And finally, a disinfection stage may be necessary to kill any remaining bacteria or microorganisms still present in the water and to protect the water as it's distributed to homes, schools and businesses.
CYANOBACTERIAL TOXINS IN DRINKING WATER
Occasionally other compounds are present in water that need to be removed before it is safe for human consumption. Blooms of toxic cyanobacteria, which used to be called blue green algae by scientists and pond scum by everyone else, are often a problem in freshwater. Some species contain toxins that cause acute health impacts such as skin irritation or chronic health impacts such as liver or kidney damage. Toxic cells need to be physically removed by sedimentation, floatation (cells are often buoyant and float at the surface) or filtration during treatment. Cells die and release their toxins into the water, so additional chemical removal steps such as adsorption on activated carbon or treatment with ozone may be necessary to remove these toxins.
 Pond scum (blue green algae or cyanobacteria). Credit: The Wine Commonsewer
Pond scum (blue green algae or cyanobacteria). Credit: The Wine CommonsewerNow that you have clear, potable (suitable for drinking) drinking water, how does it get to your tap? Water is moved to consumers through a huge network of storage tanks, valves, pumps and millions of miles of water distribution pipeline around the earth. Most of this movement occurs in infrastructure that is underground. Having the infrastructure (that network) necessary to move water from treatment to tap in place is no guarantee that water will flow from your tap unless you live at the bottom of a mountain and your water treatment plant is at the top. What's missing? In a word: Pressure. Pressure is needed to move water from point A (the treatment plant) to point B (your tap). How is water pressurized? The energy that this infrastructure needs to deliver the water (water pressure) can be transferred to water in a number of ways. The particular method used depends on a host of factors including local water needs and demands, local geography, future growth, leakage, infrastructure age, fire fighting needs and local government. Methods include pumps, compressed air and gravity feeds (from mountains, or in most cases, water towers at higher elevations) and can occur at specialized structures called pump stations.
Why is Fluoride controversial?
Fluoride is controversial for a variety of scientific, ethical and environmental reasons.
A key difference between the implementation of drinking water fluoridation in the United States and Australia was the impact of temperature and climate on water consumption. Temperatures are a key factor in the establishment of legislative requirements, such as the Water Fluoridation Regulation 2008[5] in Queensland, that prescribe concentrations of fluoride to be added to the water. Consequently, areas with higher average temperatures require less fluoride to be added to the drinking water to achieve the same oral health benefits. The tropical conditions found in parts of Australia, such as Queensland, also make it difficult to maintain fluoridation equipment due to higher levels of corrosion caused by the wet climate.
On 29 November 2012 the Queensland Parliament, with a Liberal National Party government, reversed the previous Labour government's mandate requiring certain public potable water supplies to add fluoride to the water. As a consequence of these changes local councils in Queensland have the choice to add fluoride to drinking water supplies, similar to the conditions in place under the previous legislation. In January 2013 the Cairns council decided to discontinue water fluoridation.
What is Fluoride?
- the 13th most abundant natural mineral on earth
- a heavily mined element that is present naturally in water supplies at low concentrations
- a univalent pale yellow poisonous gas at room temperature that readily forms compounds with most elements. It is the most chemically reactive and electronegative of all elements.
What is Fluoride used for?
- it is often added to toothpaste and municipal water supplies because it is elemental to producing strong bones and improves dental health. But, in large quantities, it can cause fluorosis and osteoporosis
- it is used in producing isotopically fractionated uranium (from UF6)
- it is used to make commercial fluorochemicals, including many high-temperature plastics
- hydrofluoric acid is used for etching the glass of light bulbs, etc.
- fluorochloro hydrocarbons are used extensively in air conditioning and refrigeration
Compare fluoridation attitudes in the US to Australia...your state to other states?
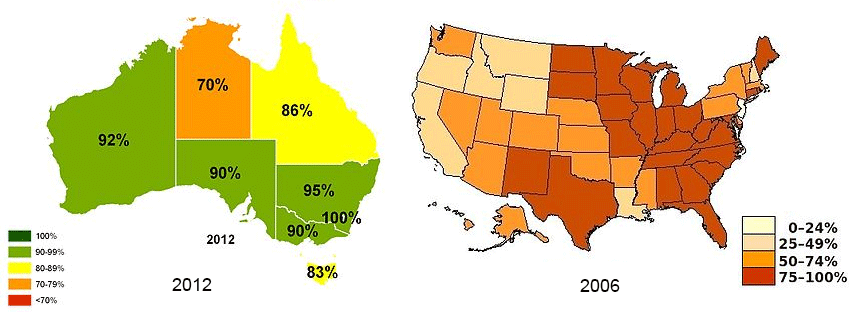 Percent coverage of water flouridation in Australia and the United States
Percent coverage of water flouridation in Australia and the United StatesHow do you feel personally about fluoridation?
TOPIC OF DISCUSSION: Why is some drinking water fluoridated? Why does fluoridation sometime cause controversy?
Scientifically: Does the evidence support its use to improve dental health? Currently, commercial toothpaste is fortified with sodium monofluorophosphate or sodium fluoride. Drinking water may be treated by the addition of one of several forms of Fluoride, including:
- hexafluorosilicic acid (H2SiF6): the most common additive, used in larger scale water treatment plants (costs more to handle, but has a lower cost)
- sodium fluoride (NaF): used by small scale water treatment plants
- sodium hexafluorosilicic acid (Na2SiF6).
HISTORY OF FLUORIDE: In the 16th century, a German physician Georg Bauer described the diseases of miners (which he ascribed to evil ghosts doing mischief in the mines) and wrote the first book on how to prepare metals from ores. He explained how a mixture of fluxes ("lapides igni liquescentes (fluores)" = lat.: "stones which become liquid in fire (flows)") helps to smelt ores, including Flußspat" (fluorspar), a fluoride compound. In 1720, it was discovered that a mixture of Fluorspar, an acid and heat results in fumes that will etch glass. From 1824-1886 many early chemists attempted to isolate fluorine, Henri Moissan succeeded in 1886.
Fluoride toothpaste and water fluoridation have been touted by some health organizations as major contributors to dental caries reduction in the United States and often cited as a public health success. The US National Institute of Health (NIH) thinks Fluoride is a huge public health success story, has saved billions of dental health care dollars by preventing tooth decay, and has spared many people the pain and suffering associated with cavities and its consequences.
Some doctors, dentists and citizens think that Fluoride additions to public water supply is having a negative impact on public health, resulting in in mottling of the teeth, lower IQ, brain damage, pineal and thyroid disfunction, kidney and reproductive problems and at high levels affecting the skeleton and ligaments and causing cancer.
What are the documented health effects of Fluoride?
In small concentrations: At the 0.7 - 1.2 ppm concentration in drinking water, fluoride increases bone density and increases calcium fluorapatite in teeth, which is generally thought to lead to fewer cavities. Fluoride regulation in drinking water supplies at the .7 - 1.2 ppm level is recommended by the American Dental Association and the World Health Organization.
With chronic exposure of higher concentrations: At higher concentrations, Fluoride has been suggested to lead to liver damage, kidney damage, and dental fluorosis. Amidst these negative findings, the American Academy of Allergy and Immunology, the American Academy of Diabetes, the American Cancer Society, the American Diabetes Association, the American Nurses Association, the American Psychiatric Association, the National Kidney Foundation, and the Society of Toxicology have discounted fluoride as a beneficial additive and no longer support its use.
Countries banning fluoride addition to drinking water: Sweden, Norway, Denmark, West Germany (now unified), Italy, Belgium, Austria, France, and The Netherlands, Japan, Italy, Iceland
Countries that don't ban fluoride addition to drinking water: US (70% of water is fluoridated), Australia, New Zealand, Ireland, U.K., Serbia, Poland and Spain
Ethically: Do we have the right to decide what to put in our own bodies? Or does the general good (and fewer cavities) outweigh the right of the individual? Some cite the US Food and Drug Administration (FDA) classification of fluoride as a drug when it is used to prevent or mitigate disease (FDA 2000) as a reason that the public needs to consent to its addition to drinking water. Informed consent is standard practice for all medication, and why many countries have ruled against fluoridation of public water.
Environmentally: Fluoride additions have environmental consequences. Once fluoride is in the environment it cannot be destroyed, it can only change form. Fluoride strongly attaches to soil and sediment particles and in soil it may accumulate in plants (depends on plant species, soil type and amount of fluorine) and cause reduced growth rate and crop yields (especially corn and apricots). Fluoride can bioaccumulate up food chain, concentrating in animals eating these fluoride containing plants, resulting in dental decay and bone degradation and low birth rates.
Reference
Kauffman, J.M. 2005. Water fluoridation: A review of recent research and actions. Journal of American Physicians and Surgeons, 10(2): 38-44.
Activity
Discuss the different perspectives decsribed in the two cartoons regarding fluoride...can you design a poster that reflects your school's collective view?

Post Comment
To leave a comment, you must be signed in








