Environmental Impacts
We have now completed the urban water cycle and discovered how we extract, treat, use and dispose of water through the urban water cycle. Unfortunately, however, we very rarely put the water back into the environment in the same condition we first received it and this can lead to a bunch of problems. “Out of sight” should not be “out of mind”. The environmental impacts of used water (wastewater) have to do with all the chemicals, solids and treatments (e.g. heating, cooling etc.) that we apply during its urban water journey that aren’t able to be fully treated prior to disposal into the environment.
Oxygen is required for all life apart from some primitive bacteria. Oxygen is produced from plant life through photosynthesis. The earth's atmosphere is approximately one-fifth oxygen and four-fifths nitrogen. Aquatic animals such as fish ‘breathe’ the atmospheric oxygen that is dissolved in water.
Probably the most known and best-studied effect of wastewater on the environment is eutrophication. Eutrophication is “the process by which a body of water becomes enriched in dissolved nutrients that stimulate the growth of aquatic plant life usually resulting in the depletion of dissolved oxygen”. Whenever oxygen gets depleted……things are not good. Unless you are an anaerobic bacteria of course!
Nutrients aren’t bad, in fact they are essential for life. The primary nutrients involved in eutrophication, nitrogen and phosphorus, are vital for healthy cellular function in all organisms (e.g. amino acid and protein production, cellular energy, genetic coding in DNA). But like everything else in life, you can have too much of a good thing.
The problems typically start with an overgrowth (or “bloom”) of microscopic plants in the water called phytoplankton. These tiny plants are very good at using nutrients to grow, multiply and take over an ecosystem. Once this happens, a cascade of events typically occur that can be unappealing to look at and difficult to stop.
 Algal blooms and fish kills.
Algal blooms and fish kills.Secondly, phytoplankton can get so thick that they limit the amount of light that penetrates the water and reaches the bottom (“benthos”). This results in unfavorable conditions for plants growing on the bottom (e.g. seagrass) that depend on sunlight for photosynthesis and growth. Resultant loss of benthic plants means loss of habitat and destabilization of the sediment which can in turn be stirred up by waves/wind making the light and nutrient problem worse.
Thirdly, phytoplankton can reach such numbers that they run out of resources (i.e. food and light) to sustain themselves and begin to die and sink to the bottom. This is where bacteria begin to “bloom” and start the decomposition of the huge quantities of dead phytoplankton, and the benthic plants that have died due to a lack of light. Bacteria do not photosynthesize or produce oxygen like phytoplankton, rather they use oxygen (“respire”) and can very quickly use up all the oxygen in the water resulting in the mass death of aquatic organisms (e.g. fish kill).
All of this if often associated with a bad smell, degraded aesthetics and very unhappy local citizens!
The eutrophication scenario just described is one possible cascade of effects that can result from excessive nutrients into the aquatic environment. There are a number of other famous eutrophication cases. Do you know of any? (Hint: Kanahoe Bay, Hawaii; and more recently on the beaches in China. Whoa!). Also, how do you think a phytoplankton bloom would affect your drinking water??
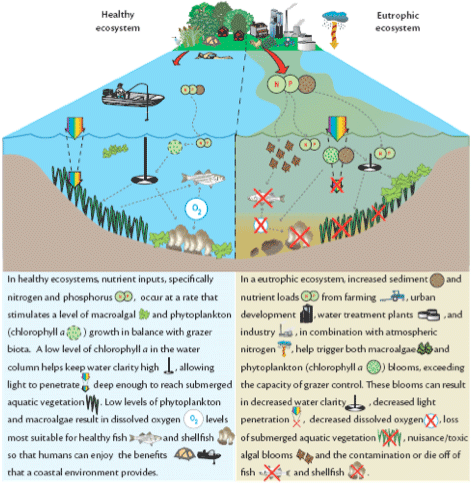 Conceptual diagram showing the impacts of eutrophication.
Conceptual diagram showing the impacts of eutrophication.
Many industrial wastewaters contain dissolved metals as a result of their manufacturing processes. Metals such as arsenic, mercury, lead, cadmium, aluminum, and iron end up in the wastewater from many industries such as manufacturing and mining. Most governments have laws in place that govern how much of these metals are allowed to be in the wastewater. However, even small amounts of heavy metals can have negative impacts to fish and other animals that live in the waterways, including reduced growth and development, cancer, organ damage, and nervous system damage.
 Pharmaceuticals and personal care products
Pharmaceuticals and personal care productsMonitoring
There often is a requirement for those responsible for generating wastewater to conduct or fund monitoring programs to assess water quality and those organisms that depend on a clean and healthy ecosystem. This may be an industry or could be you through your government, funded by taxes. Monitoring is required to understand environmental impacts from wastewater, and ensure that wastewater is being treated as required and that the ecosystem is not being degraded beyond what has been determined acceptable (what do you think “acceptable degradation” means?).
There are many ways to monitor water quality, analyze the results, and present them in a meaningful way. Here are a couple of examples of some of the worlds biggest water quality monitoring programs:
- The Healthy Waterways Ecosystem Health Monitoring Program in Southeast Queensland, Australia, monitors water quality in the region’s rivers and bays. They monitor 135 freshwater sites twice per year, and 254 estuarine and marine sites every month.
- The Chesapeake Bay Program monitors important habitats, fish and shellfish, and water quality measures in Chesapeake Bay in the USA, as these are good indicators of the overall health of Chesapeake Bay and the animals that live in it.
Each program monitors a wide range indicators relevant to what is considered a healthy ecosystem in each location. What makes these monitoring programs so successful, is not that they collect huge amounts of information, but that they synthesize all the information into an annual ecosystem health report card that communicates the status of the local ecosystem to the public and how it is changing over time.
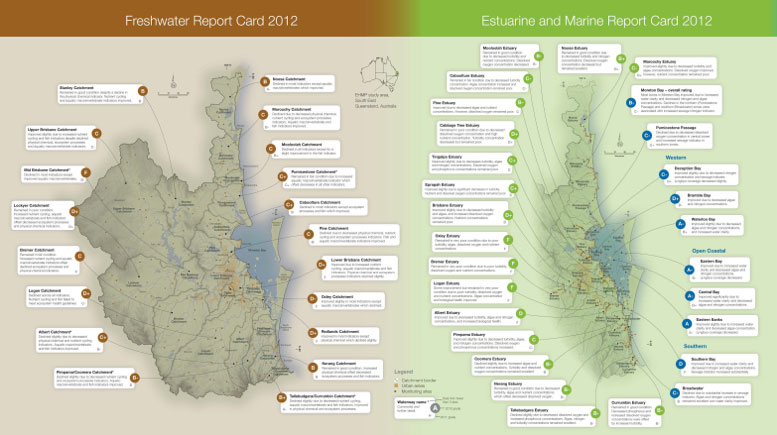 Ecosystem Health Monitoring Program report cards.
Ecosystem Health Monitoring Program report cards.
Post in the comments below what issues arising from wastewater, if any, are affecting your local waterways and what is being done about it. Provide links to data, reports or websites that present the data.
Post Comment
To leave a comment, you must be signed in


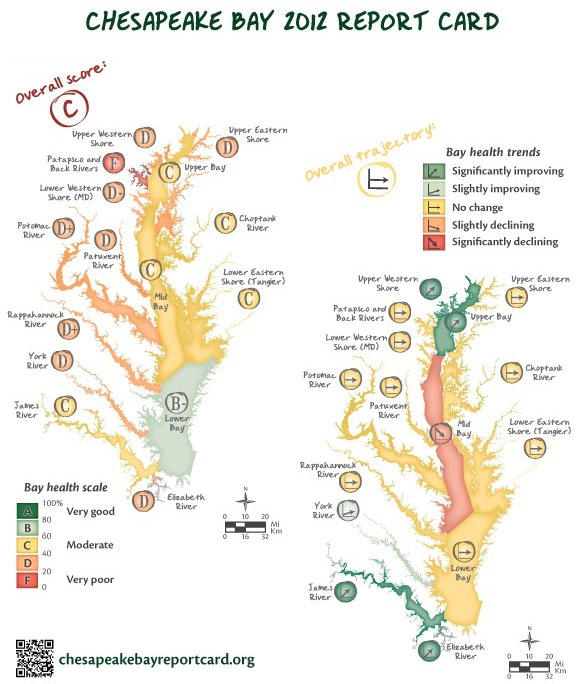 Chesapeake Bay Report Card
Chesapeake Bay Report Card





